Case Summary
- Ms O’Riordan is in her late 70s and has very inflamed joints of her hands.
- Two weeks after our initial visit to the O’Riordan house, Ms O’Riordan admits to pain, parasthesia, and loss of strength in her hands, and particularly her thumb.
- Assuming the median nerve may be impacted by the inflammation, we suggest a minor operation to free the nerve, but O’Riordan is hesitant and concerned about the risks involved.
- While O’Riordan contemplates the surgery, we are reviewing her drug regime to see if we can decrease the swelling by that route.
Identify the major skeletal structures of the forearm and wrist.
Review of bone modelling
- Bone growth - cartilage proliferates, are calcified, are dissolved by osteoclasts, then osteoblasts move in and lay down collagen, turn into osteocytes, and form bone.
- Endochondral ossification
- Mesenchymal cells develop into a hyaline cartilage model of the future bone
- Primary ossification center forms at future diaphysis and cartilage is converted to bone.
- Secondary ossification center forms at the epiphysis of the bone
- Growth in length involves two events:
- Growth of cartilage on the epiphyseal side of the epiphyseal plate
- Replacement of cartilage on the diaphyseal side of the epiphyseal plate with bone by endochondral ossification
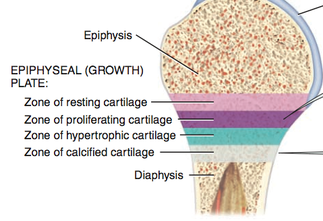
- The epiphyseal plate is a layer of cartilage in the metaphysis that contains 4 zones, in order of closest to epiphysis
- Zone of resting cartilage
- Cartilage just relaxing in the epiphyseal plate and do not function in bone growth
- Zone of proliferating cartilage
- Cartilage that is dividing and growing to replace those that die at the diaphyseal side
- Zone of hypertrophic cartilage
- Mature cartilage
- Zone of calcified cartilage
- Extracellular matrix around the cartilage calcifies the cartilage and the cartilage dies and are dissolved by osteoclasts. Osteoblasts and capillaries from the diaphysis then invade the area and osteoblasts secrete collagen to create bone extracellular matrix, trapping themselves inside, become osteocytes, and bone is formed.
- The cartilage has been transformed to bone, and is now part of the diaphysiss
- Extracellular matrix around the cartilage calcifies the cartilage and the cartilage dies and are dissolved by osteoclasts. Osteoblasts and capillaries from the diaphysis then invade the area and osteoblasts secrete collagen to create bone extracellular matrix, trapping themselves inside, become osteocytes, and bone is formed.
- Zone of resting cartilage
- Ongoing replacement of old bone by osteoclasts and osteoblasts à PTH promotes bone resorption, calcitonin promotes bone deposition.
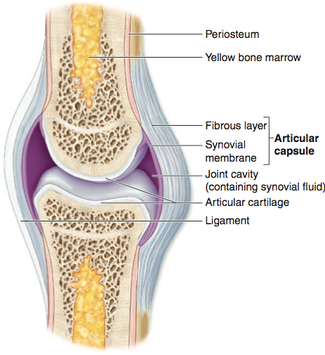
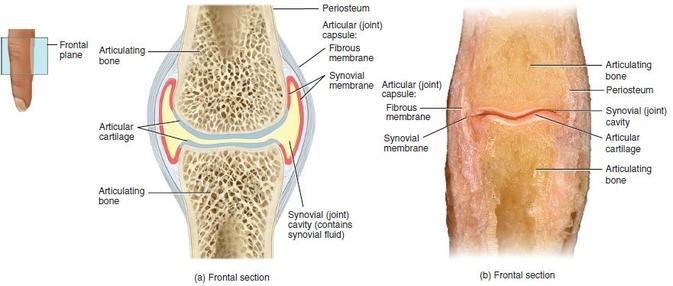
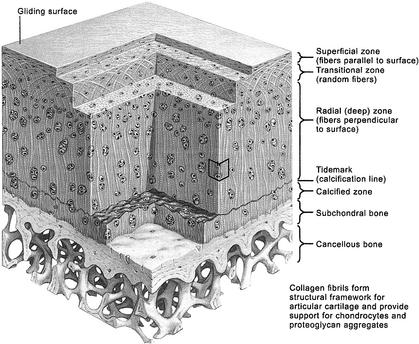
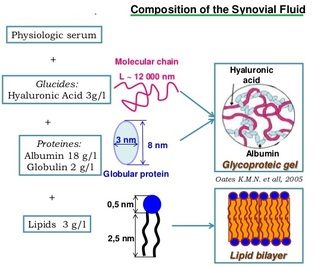
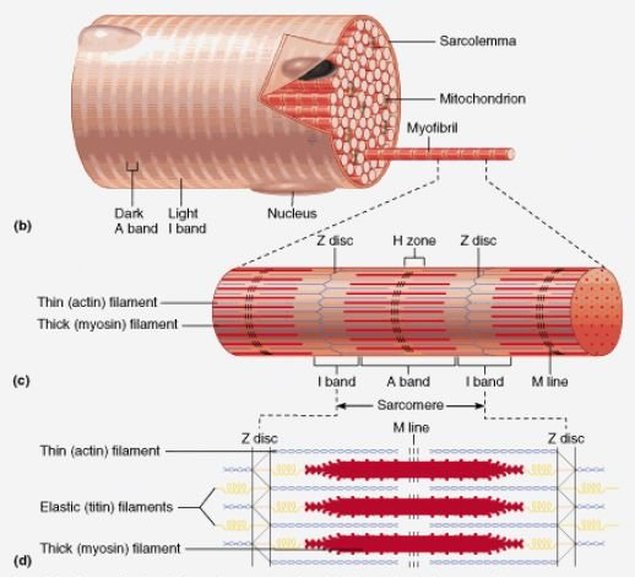
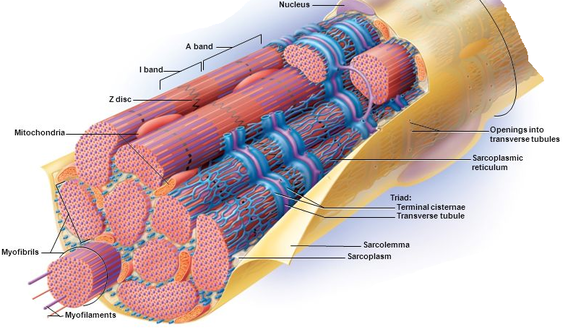
Cartilage |
Synovial Fluid |
|
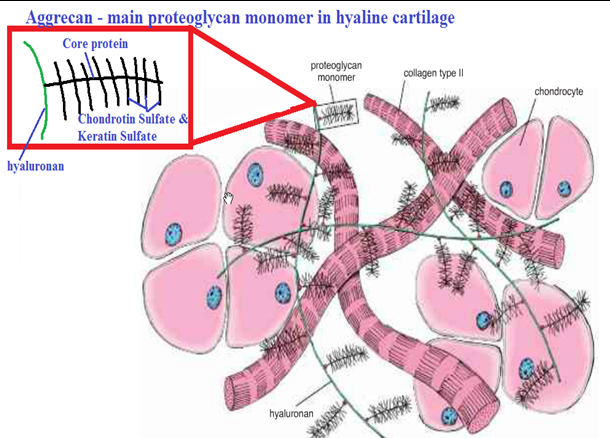
Cartilage is connective tissue that consists of collagen and elastic fiber, and like other CT it has few cells and large quantities of ECM. It differs from other CT though by not having any nerves or blood supply.
|
|
Identify the muscles of the forearm and hand and describe their functions and innervation.
Muscles of the forearm and hand, and their function and innervation
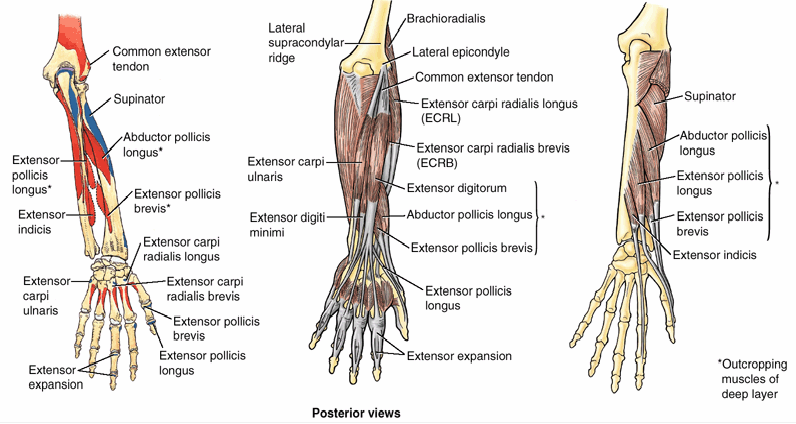
Forearm
Generally the flexor-pronator muscles attach to medial epicondyle and spiral anteriorly, while extensor-supinator muscles attach to lateral epicondyle and spiral posteriorly.
Forearm
Generally the flexor-pronator muscles attach to medial epicondyle and spiral anteriorly, while extensor-supinator muscles attach to lateral epicondyle and spiral posteriorly.
- Flexors and pronators are supplied by the median nerve (other than the flexor carpi ulnaris and the medial half of the flexor digitorum profundus)
- Extensors and supinators are all supplied by the radial nerve
- Superficial layer – attach to medial epicondyle by common flexor tendon
- Pronator teres
- Secondary pronator
- Palmaris longus
- Flexion at wrist
- Flexor carpi radialis
- Flexion and abduction of the wrist
- Flexor carpi ulnaris (ulnar nerve)
- Flexion and adduction of wrist
- Pronator teres
- Intermediate layer
- Flexor digitorum superficialis
- Flexes the middle phalanges of the medial 4 fingers at the proximal interphalangeal joints, as well as proximal phalange in continued action
- Can flex all 4 fingers independently
- Flexor digitorum superficialis
- Deep layer – the deeper you go the further you get
- Flexor digitorum profundus (medial ½ ulnar nerve)
- Flexes the distal phalanges of medial 4 fingers after FDS has flexed middle phalanges.
- Can only flex the index finger independently
- Flexor pollicis longus
- Long flexor of the thumb
- Pronator quadratus
- Primary pronator
- Flexor digitorum profundus (medial ½ ulnar nerve)
- One major exception – the brachioradialis is functionally a flexor of the forearm at elbow, but it is in the posterior (extensor) compartment and is supplied by radial nerve.
- Long flexors of the digits also flex the MCP and wrist joint
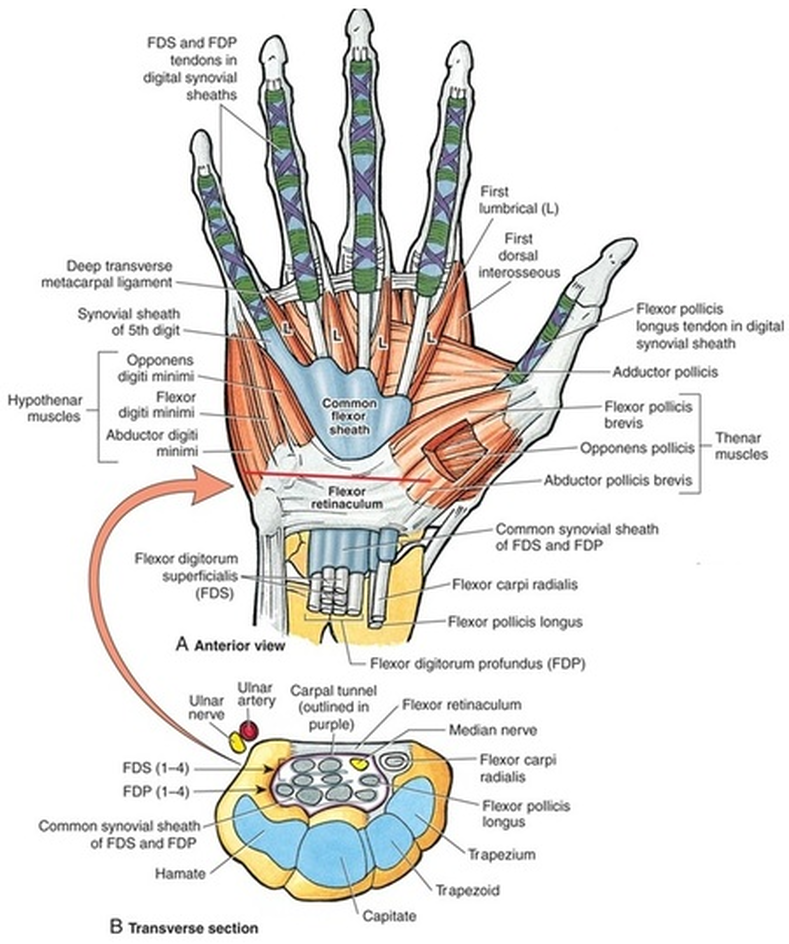
- The tendons of the flexors are held in place distally by the palmar carpal ligament and the flexor retinaculum
|
Extensor-supinator muscles
Muscles that act on wrist joint
|
Intrinsic muscles of hand
Compartments of the hand 1. Thenar muscles in the thenar compartment (median nerve) Abductor pollicis brevis
2. Adductor compartment (ulnar nerve) Adductor pollicis
Abductor digiti minimi
1st and 2nd lumbricals are median nerve, 3rd and 4th are ulnar
|
Describe the structure and movement of the joints of the forearm, wrist and hand.
Forearm
Elbow joint
Elbow joint
- Hinge type synovial joint between the humerus, ulna, and radius. 3 articulations:
- The trochlea of the humerus articulates with the trochlear notch of the ulna (formed by the olecranon and coronoid process)
- Capitulum of the humerus articulates with the concave head of the radius
- The radius articulates with the ulna at the proximal radio-ulnar joint, which is a pivot synovial joint (head of radius into radial notch of ulna).
- Ligaments of the elbow
- Ulnar collateral ligament
- On medial side, 3 bands extend from the medial epicondyle of the humerus to the coronoid process and olecranon of the ulna
- Radial collateral ligament
- From lateral epicondyle of the humerus and blends distally with the anular ligament of the radius.
- Anular ligament
- Holds the head of the radius in the radial notch of the ulna to form the proximal radio-ulnar joint.
- Ulnar collateral ligament
- Movements at the elbow joint
- Flexion and extension at the elbow hinge joint
- Flexors – brachialis, brachioradialis and biceps brachii
- Extensor – triceps brachii
- Proximal radio-ulnar joint permits pronation and supination of the forearm
- Supination – supinator and biceps brachii
- Pronation – pronator quadratus (primarily) and pronator teres
- Flexion and extension at the elbow hinge joint
- Is a pivot joint
- Head of ulna articulates with ulnar notch of radius
- Ends of the ulna and radius bound together by a fibrocartilaginous articular disc that goes from the medial edge of the radius to the medial edge of the ulna
- Ligaments
- Transverse ligaments extend from radius to ulna on the anterior and posterior side to strengthen joint.
- Movement
- Pronation and supination – radius moves around the fixed distal end of ulna
|
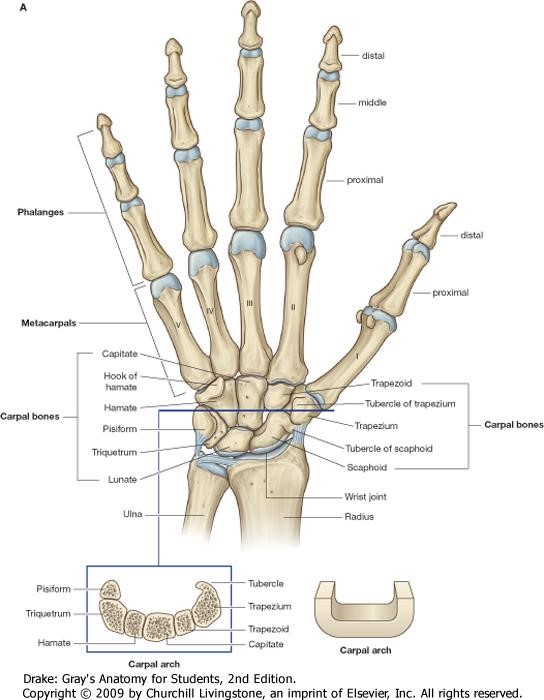
Wrist - Bones
8 carpal bones arranged in two rows of 4
|
Intercarpal joints
- Interconnecting the carpal bones are plane synovial joints between carpal bones of the proximal row, carpal bones of the distal row, midcarpal joint between the proximal and distal row, and the pisotriquetral joint.
- Ligaments
- Carpals are united by anterior, posterior, and interosseus ligaments.
- Movements
- Gliding between carpals occurs with movements at wrist joint
- Flexion and extension of hand are initiated at midcarpal joint
Hand
Bones
Bones
- The distal row of carpal bones articulate with the metacarpals. There are 5 metacarpals, which form the palm of the hand.
- The distal end of the metacarpals articulate with the proximal phalanges and form the knuckles.
- Each finger has three phalanges, except the thumb which has 2. The proximal are largest, middle are intermediate, distal are smallest.
- Are plane type synovial, except for the carpometacarpal joint of the thumb, which is saddle
- Occurs between the distal row of the carpus and the base of the metacarpals.
- The CMC joint of the thumb is between the trapezium and the base of the 1st metacarpal
- Adjacent metacarpals articulate with eachother at the base of the metacarpals
- Ligaments of CMC and IM
- Palmar and dorsal carpometacarpal and intermetacapral ligaments
- Interosseus intermetacarpal ligaments
- Transverse metacarpal ligaments between the distal ends of the metacarpals
- Movement of CMC and IM
- CMC joint of thumb allows for movement in any plane (F/E, Ab/Ad, circ, opp)
- Almost no movement at CMC of 2nd and 3rd finger, 4th is slightly more mobile, and 5th moderately mobile.
- MCP are condyloid synovial joints
- IP joints are hinge type joints
- Ligaments
- Each MCP and IP joint is strengthened by two (medial and lateral) collateral ligaments that have two parts:
- Cord like part – go from head of metacarpal and phalanges to distal phalanges
- Are slack during extension, taut during flexion
- Fan like part – pass anteriorly and attach to palmar ligaments of CMC
- Cord like part – go from head of metacarpal and phalanges to distal phalanges
- Each MCP and IP joint is strengthened by two (medial and lateral) collateral ligaments that have two parts:
- Movement
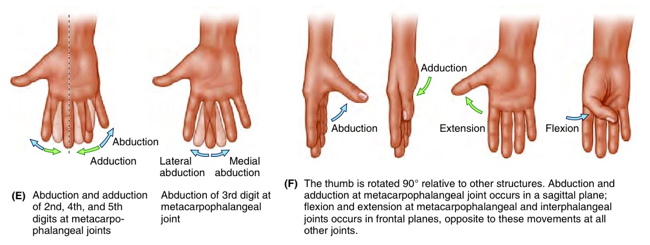
- At 2nd to 5th digit, metacarpophalageal joint allows F/E, Ad/Ab, and circ
- MCP joint of thumb limited to flexion and extension
- At IP joints, only flexion and extension
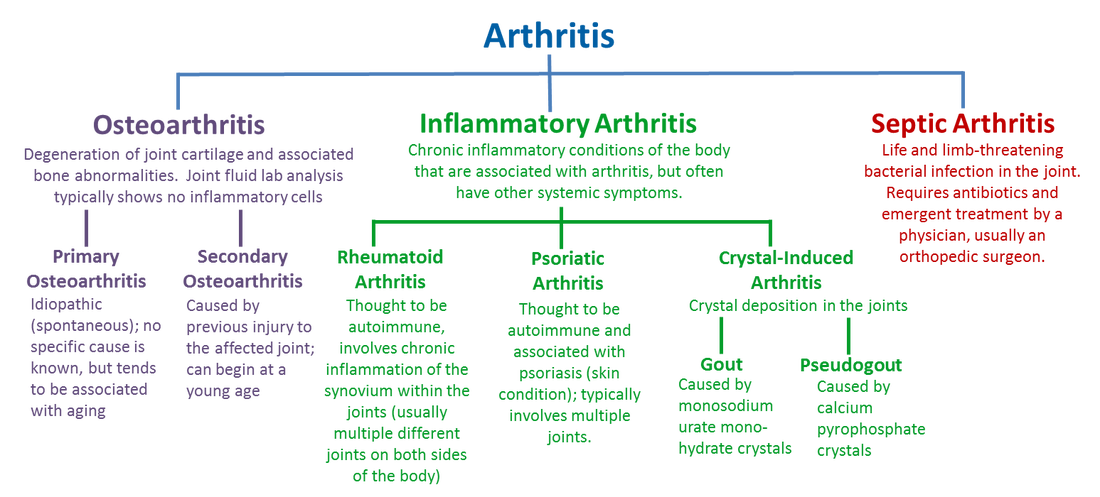
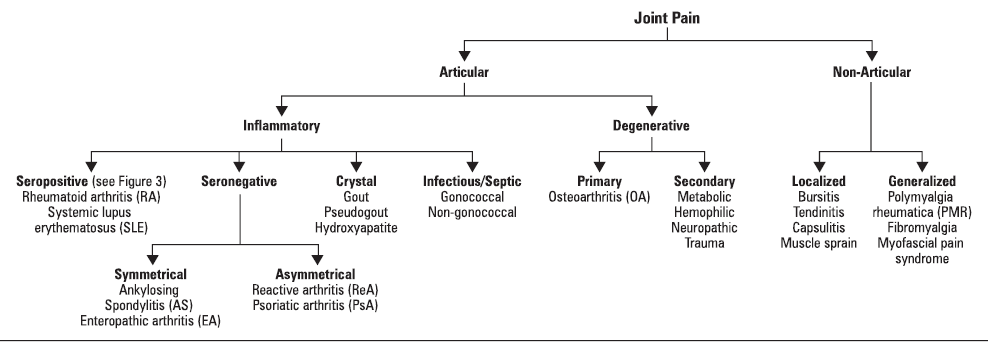
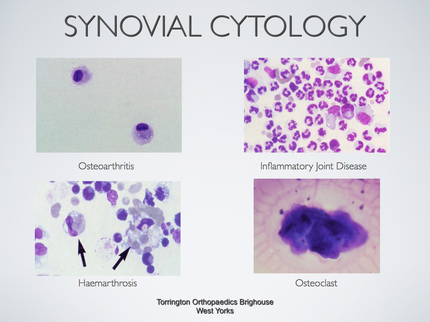
Compare RA with other common inflammatory joint diseases such as gout.
|
Rheumatoid arthritis
|
Osteoarthritis
|
Gout
- Recurring attacks of acute inflammatory arthritis
- Pathophysiology
- Elevated levels of uric acid in the blood crystalize and deposit in joint, inciting inflammation
- High uric acid due to rich diet, genetic predisposition, or under excretion of uric acid
- Rich diet – alcohol, sweetened drink, red meat, seafood (high purine)
- High uric acid due to rich diet, genetic predisposition, or under excretion of uric acid
- Elevated levels of uric acid in the blood crystalize and deposit in joint, inciting inflammation
- Clinical features
- Reoccurring attack of inflammation, red, painful, hot, swollen joint
- Most commonly found in the metatarsal-phalangeal joint of big toe
- Gout commonly occurs in combination with obesity, hypertension, or use of diuretics
- Fatigue and fever
Describe the main components of the development of inflammatory joint disease and its associated pathology, using rheumatoid arthritis as an example.
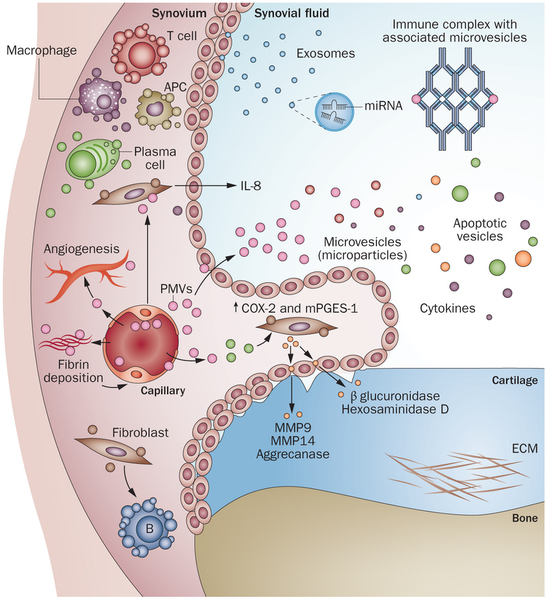
- Synovitis – inflammation of the synovial membrane of joints leading to pain, swelling, redness, and nodules in the joint. The joint will swell due to synovial fluid collection. Long term synovitis can result in degeneration of the joint.
- RA is an autoimmune disease that is caused by a combination of factors including genetic factors and environmental factors, which contribute to the breakdown of self tolerance by the immune system causing synovial inflammation.
- The pathological changes are caused mainly by CD4 T Helper cells, which are inappropriately activated and release cytokines that result in inflammation.
- T helper cell may be activated by a microbe or self antigen
- Activated T helper cells release cytokines (IL-1 and TNF) that recruit leukocytes, activate resident synovial cells, activate B cells, and activate osteoclasts.
- Macrophages will come in and release toxic agents that cause tissue injury
- Macrophages and T helper cells activate fibroblasts
- Synovial cells will be promoted to proliferate (leads to thickened villous folds) and produce proteolytic enzymes like collagenase that will destroy cartilage, ligaments, and tendons of the joint.
- There is subsequent angiogenesis to nourish the influx of cells and activity
- B-cells are activated to produce IgM autoantibodies (ie rheumatoid factor)
- IgM autoantibodies bind the Fc portion of IgG and form immune complexes that deposit in joints and other tissues causing inflammation and tissue damage.
- Activated T helper cells release RANKL which increase osteoclast activity and erode bone.
- Cause the osteopenia
- Macrophages will come in and release toxic agents that cause tissue injury
- These actions create a pannus – a mixture of proliferating synovial cells, inflammatory cells, granulation tissue, and fibrous connective tissue
- The articular cartilage next to the pannus is eroded, and eventually the joint space is filled by the pannus leading to fibrosis and ossification leading to permanent ankylosis (immobility of a joint due to fusion of the bones)
- Rheumatoid nodules can develop on the arm, or in the lungs, heart etc. They are characterized by a central fibrinoid necrosis surrounded by macrophages which are surrounded by granulation tissue and lymphocytes
- Patients with nodules and high rheumatoid factor are also at risk of developing vasculitis
- TNF and IL-1 antagonist
- X-ray of hand and feet
- Osteopenia, loss of joint space, bony erosions, subluxation
- Blood test
- Test for RF – not having RF does not rule RA out
- Erythrocyte sedimentation rate – rate at which red blood cells sediment in one hour
- Measure of inflammation, since in inflammation high proportion of fibrinogen in the blood causes RBC to stick together in stacks called rouleaux which settle faster.
- C-reactive protein – rises in inflammation
- Full blood count
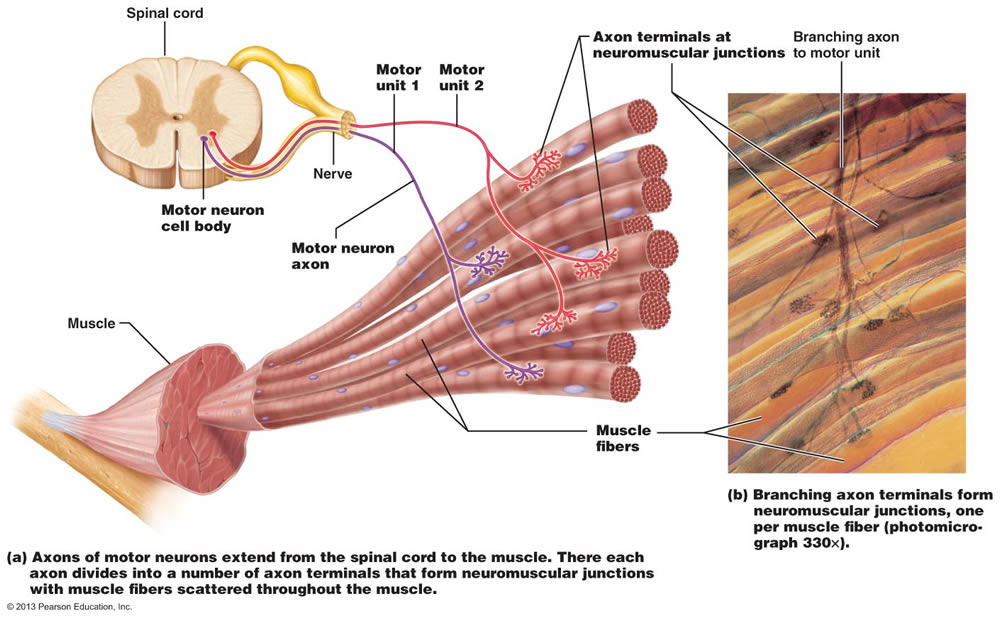
Describe the basic organisation of motor units and their relevance to the fine control and strength of muscle activity.
|
Motor Units
Motor Unit = Single motorneuron and the group of muscle fibres innervated by it. Each whole muscle is innervated by a number of different motorneurons.
|
- Two muscle fibers adjacent to each other can be connected to different motor neurons and thus form different motor units
- Force is dispersed across a muscle (over many random muscle fibers) which strengthens the contraction; thus this random dispersion of motor units is very beneficial
- The number of muscle fibers per motor unit in the gastrocnemius muscle (for example) is huge, but there are only a small number of motor units (this is the case for powerful muscles)
- In contrast, a muscle that is involved in precision and fine movements (such as the lateral rectus muscle of the eye) will have a large number of motor units but a small number of muscle fibers per motor unit
- To increase force you need to recruit (switch on) more motor units
- Slow-firing motor units (type I slow twitch fibers) are recruited first followed by quick-firing motor units (type II fast twitch); thus, fatigue resistant motor units are recruited first
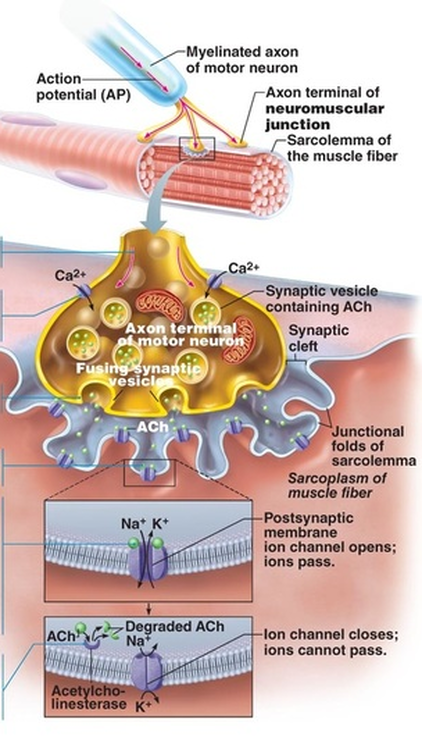
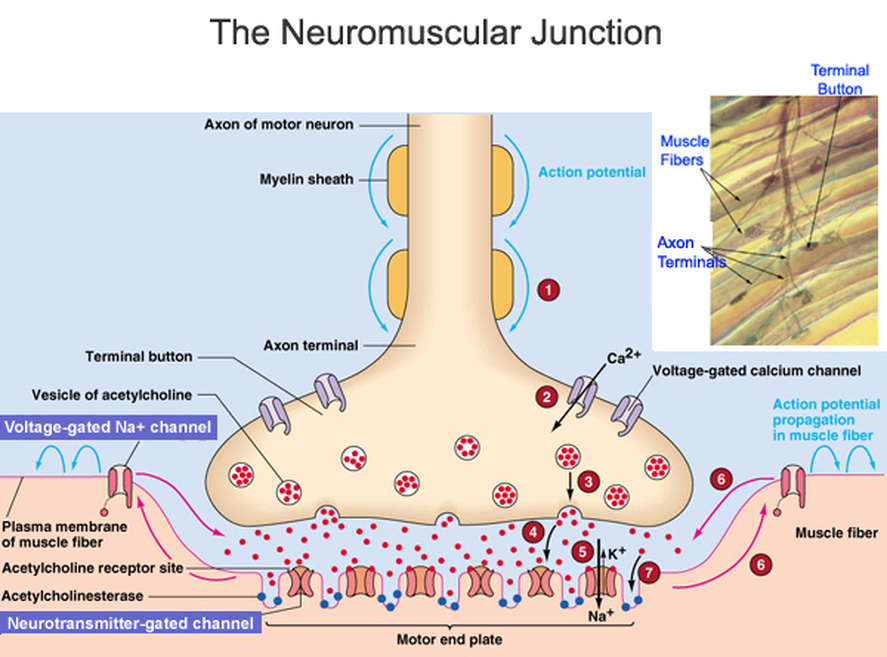
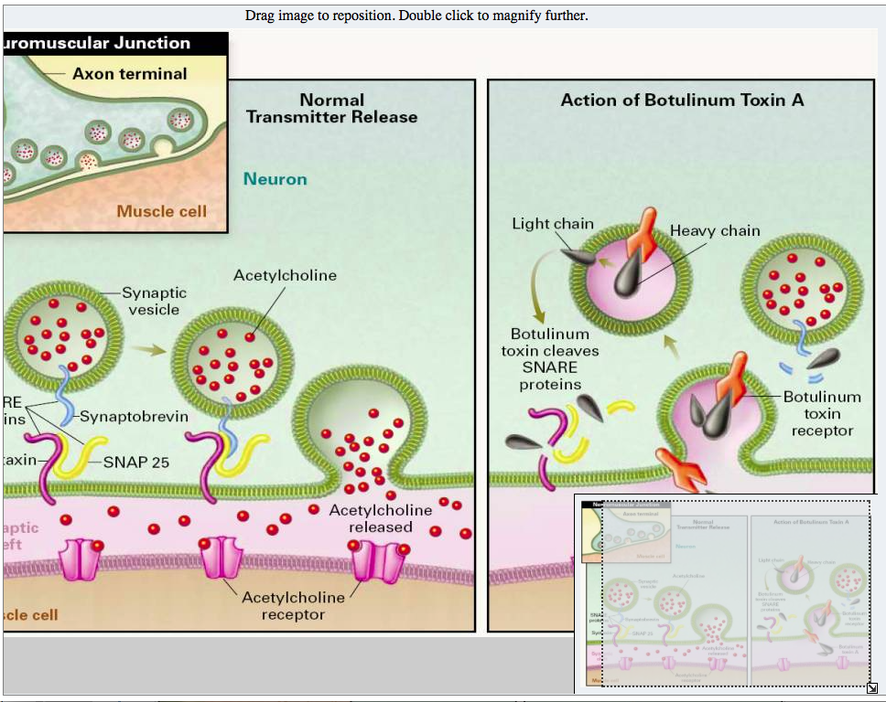
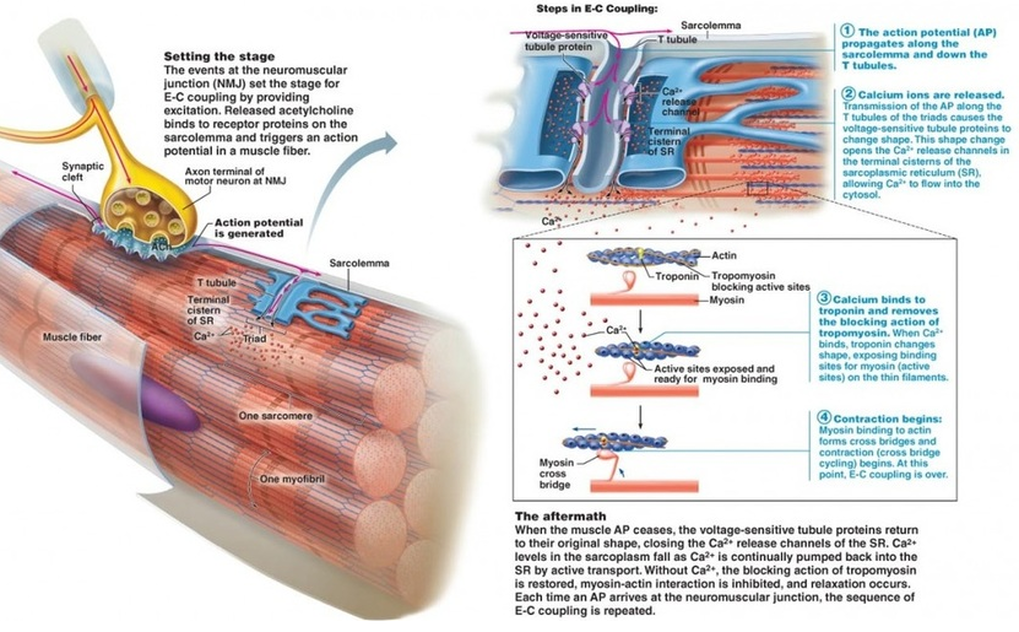
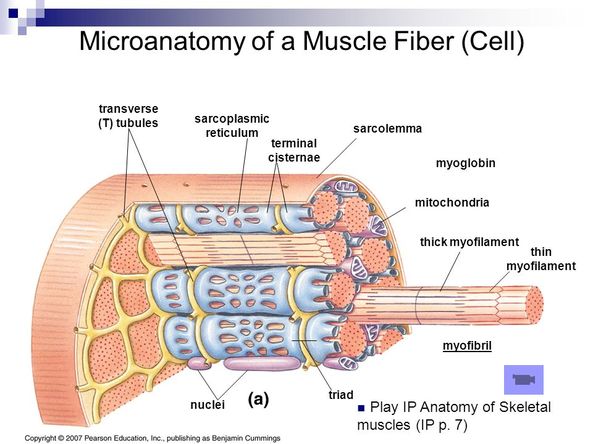
Basic processes of neuromuscular transmission and muscle contraction.
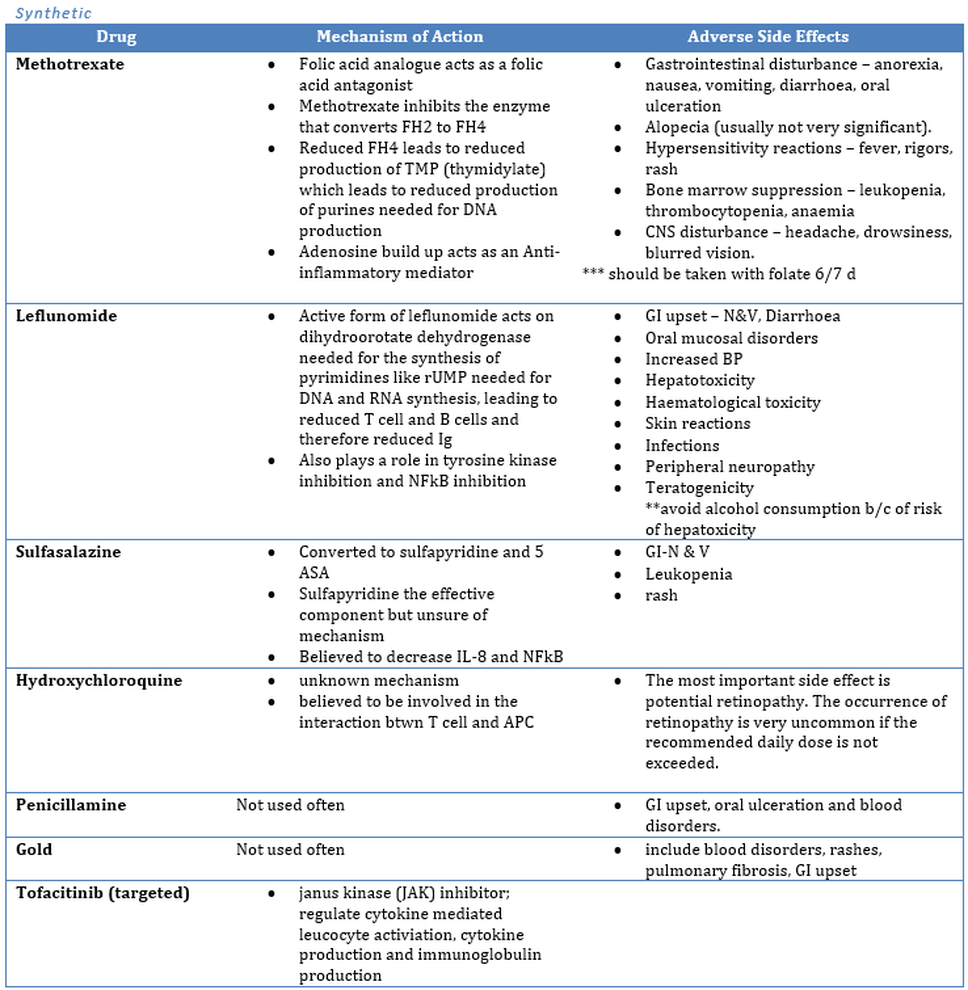
Conventional disease-modifying anti-rheumatic drugs. (DMARDS)
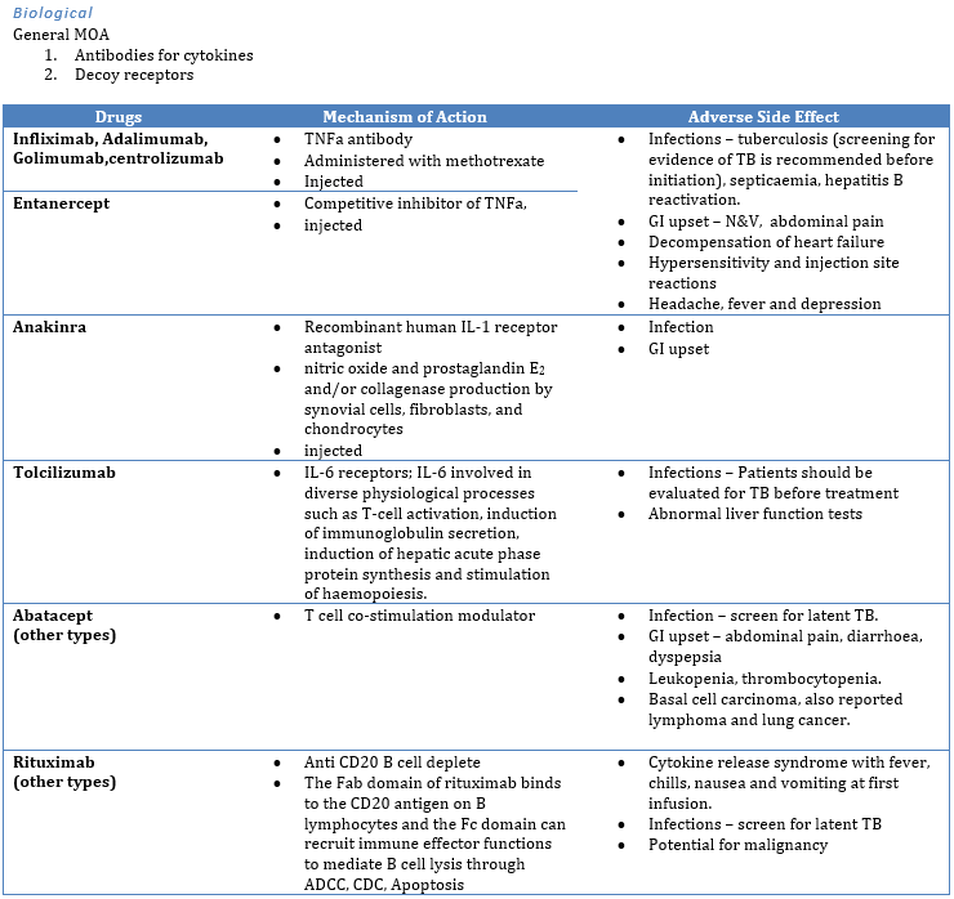
Biological anti-arthritis drugs
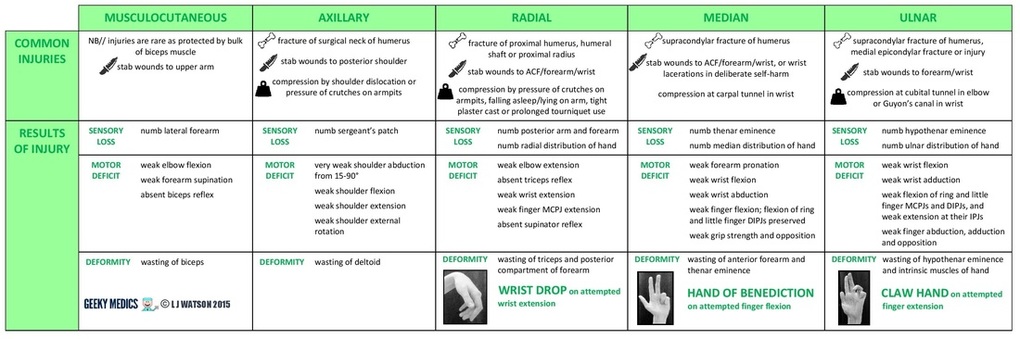
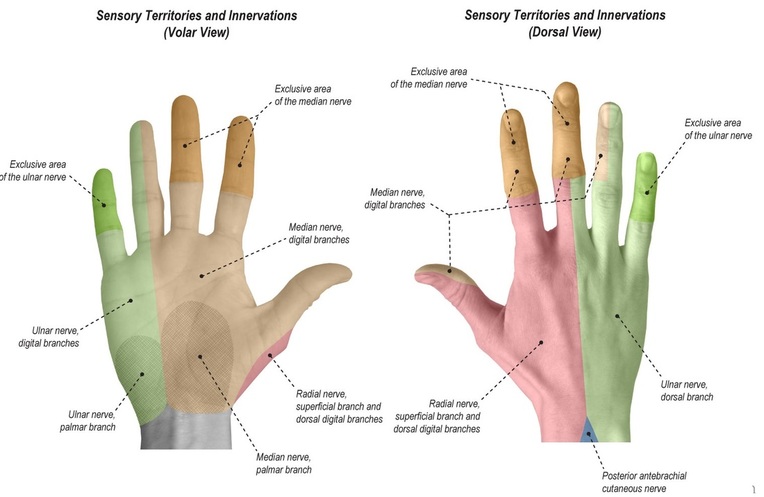
Main consequences of major nerve injury on the functions of the forearm and hand.
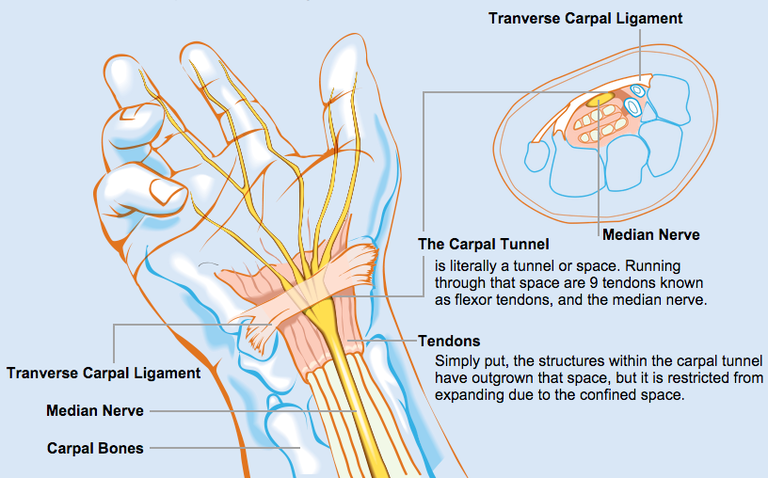
Pathophysiology and treatment of carpal tunnel syndrome and the provocation tests for carpal tunnel syndrome.
Physical exam
Phalen's maneuver is performed by flexing the wrist gently as far as possible, then holding this position and awaiting symptoms. A positive test is one that results in numbness in the median nerve distribution when holding the wrist in acute flexion position within 60 seconds. The quicker the numbness starts, the more advanced the condition. Phalen's sign is defined as pain and/or paresthesias in the median-innervated fingers with one minute of wrist flexion.
Tinel's sign, a classic — though less sensitive - test is a way to detect irritated nerves. Tinel's is performed by lightly tapping the skin over the flexor retinaculum to elicit a sensation of tingling or "pins and needles" in the nerve distribution. Tinel's sign (pain and/or paresthesias of the median-innervated fingers with percussion over the median nerve) is less sensitive, but slightly more specific than Phalen's sign
Durkan test, carpal compression test, or applying firm pressure to the palm over the nerve for up to 30 seconds to elicit symptoms has also been proposed.
Hand elevation test The hand elevation test has higher sensitivity and specificity than Tinel's test, Phalen's test, and carpal compression test. Chi-square statistical analysis confirms the hand elevation test is not ineffective compared with Tinel's test, Phalen's test, and carpal compression test
Phalen's maneuver is performed by flexing the wrist gently as far as possible, then holding this position and awaiting symptoms. A positive test is one that results in numbness in the median nerve distribution when holding the wrist in acute flexion position within 60 seconds. The quicker the numbness starts, the more advanced the condition. Phalen's sign is defined as pain and/or paresthesias in the median-innervated fingers with one minute of wrist flexion.
Tinel's sign, a classic — though less sensitive - test is a way to detect irritated nerves. Tinel's is performed by lightly tapping the skin over the flexor retinaculum to elicit a sensation of tingling or "pins and needles" in the nerve distribution. Tinel's sign (pain and/or paresthesias of the median-innervated fingers with percussion over the median nerve) is less sensitive, but slightly more specific than Phalen's sign
Durkan test, carpal compression test, or applying firm pressure to the palm over the nerve for up to 30 seconds to elicit symptoms has also been proposed.
Hand elevation test The hand elevation test has higher sensitivity and specificity than Tinel's test, Phalen's test, and carpal compression test. Chi-square statistical analysis confirms the hand elevation test is not ineffective compared with Tinel's test, Phalen's test, and carpal compression test
Review the pharmacology of non-steroidal anti-inflammatory drugs.
Medical usesNSAIDs are usually used for the treatment of acute or chronic conditions where pain and inflammation are present.
NSAIDs are generally used for the symptomatic relief of the following conditions:
- Osteoarthritis
- Rheumatoid arthritis
- Mild-to-moderate pain due to inflammation and tissue injury
- Low back pain
- Inflammatory arthropathies (e.g., ankylosing spondylitis, psoriatic arthritis, reactive arthritis)
- Tennis elbow
- Headache
- migraine
- Acute gout
- Dysmenorrhoea (menstrual pain)
- Metastatic bone pain
- Postoperative pain
- Muscle stiffness and pain due to Parkinson's disease
- Pyrexia (fever)
- Ileus
- Renal colic
- They are also given to neonate infants whose ductus arteriosus is not closed within 24 hours of birth
ContraindicationsNSAIDs may be used with caution by people with the following conditions:
- Irritable bowel syndrome
- Persons who are over age 50, and who have a family history of GI(gastrointestinal) problems
- Persons who have had past GI problems from NSAID use
- Peptic ulcer or stomach bleeding
- Uncontrolled hypertension
- Kidney disease
- People that suffer with inflammatory bowel disease (Crohn's disease or ulcerative colitis)
- Past transient ischemic attack (excluding aspirin)
- Past stroke (excluding aspirin)
- Past myocardial infarction (excluding aspirin)
- Coronary artery disease (excluding aspirin)
- Undergoing coronary artery bypass surgery
- Taking aspirin for heart
- In third trimester of pregnancy
- Persons who have undergone gastric bypass surgery
- Persons who have a history of allergic or allergic-type NSAID hypersensitivity reactions, e.g. aspirin-induced asthma
The widespread use of NSAIDs has meant that the adverse effects of these drugs have become increasingly common. Use of NSAIDs increases risk of having a range of gastrointestinal (GI) problems. When NSAIDs are used for pain management after surgery they cause increased risk of kidney problems.
An estimated 10–20% of NSAID patients experience dyspepsia. In the 1990s high doses of prescription NSAIDs were associated with serious upper gastrointestinal adverse events, including bleeding. Over the past decade, deaths associated with gastric bleeding have declined.
NSAIDs, like all drugs, may interact with other medications. For example, concurrent use of NSAIDs and quinolones may increase the risk of quinolones' adverse central nervous system effects, including seizure.
Combinational risk[edit]If a COX-2 inhibitor is taken, a traditional NSAID (prescription or over-the-counter) should not be taken at the same time. In addition, people on daily aspirin therapy (e.g., for reducing cardiovascular risk) must be careful if they also use other NSAIDs, as these may inhibit the cardioprotective effects of aspirin.
Rofecoxib (Vioxx) was shown to produce significantly fewer gastrointestinal adverse drug reactions (ADRs) compared with naproxen. This study, the VIGOR trial, raised the issue of the cardiovascular safety of the coxibs. A statistically significant increase in the incidence of myocardial infarctions was observed in patients on rofecoxib. Further data, from the APPROVe trial, showed a statistically significant relative risk of cardiovascular events of 1.97 versus placebo[21]—which caused a worldwide withdrawal of rofecoxib in October 2004.
Use of methotrexate together with NSAIDS in rheumatoid arthritis is safe, if adequate monitoring is done.
CardiovascularNSAIDs aside from aspirin, both newer selective COX-2 inhibitors and traditional anti-inflammatories, increase the risk of myocardial infarction and stroke.[23][24] They are not recommended in those who have had a previous heart attack as they increase the risk of death and/or recurrent MI. Evidence indicates that naproxen may be the least harmful out of these.
NSAIDs aside from (low-dose) aspirin are associated with a doubled risk of heart failure in people without a history of cardiac disease. In people with such a history, use of NSAIDs (aside from low-dose aspirin) was associated with a more than 10-fold increase in heart failure. If this link is proven causal, researchers estimate that NSAIDs would be responsible for up to 20 percent of hospital admissions for congestive heart failure. In people with heart failure, NSAIDs increase mortality risk (hazard ratio) by approximately 1.2–1.3 for naproxen and ibuprofen, 1.7 for rofecoxib and celecoxib, and 2.1 for diclofenac.
On 9 July 2015, the FDA toughened warnings of increased heart attack and stroke risk associated with nonsteroidal anti-inflammatory drugs (NSAID). Aspirin is an NSAID but is not affected by the new warnings.[29]
Possible erectile dysfunction risk. A 2005 Finnish study linked long term (over 3 months) use of NSAIDs with an increased risk of erectile dysfunction. This study was correlational only, and depended solely on self-reports (questionnaires).
A 2011 publication in the Journal of Urology received widespread publicity. According to this study, men who used NSAIDs regularly were at significantly increased risk of erectile dysfunction. A link between NSAID use and erectile dysfunction still existed after controlling for several conditions. However, the study was observational and not controlled, with low original participation rate, potential participation bias, and other uncontrolled factors. The authors warned against drawing any conclusion regarding cause.
Gastrointestinal. The main adverse drug reactions (ADRs) associated with NSAID use relate to direct and indirect irritation of the gastrointestinal (GI) tract. NSAIDs cause a dual assault on the GI tract: the acidic molecules directly irritate the gastric mucosa, and inhibition of COX-1 and COX-2 reduces the levels of protective prostaglandins. Inhibition of prostaglandin synthesis in the GI tract causes increased gastric acid secretion, diminished bicarbonate secretion, diminished mucus secretion and diminished trophic effects on epithelial mucosa.
Common gastrointestinal ADRs include:
- Nausea/vomiting
- Dyspepsia
- Gastric ulceration/bleeding
- Diarrhea
Ulceration risk increases with therapy duration, and with higher doses. To minimise GI ADRs, it is prudent to use the lowest effective dose for the shortest period of time—a practice that studies show is often not followed. Recent studies show that over 50% of patients who take NSAIDs have sustained some mucosal damage to their small intestine.
There are also some differences in the propensity of individual agents to cause gastrointestinal ADRs. Indomethacin, ketoprofen andpiroxicam appear to have the highest prevalence of gastric ADRs, while ibuprofen (lower doses) and diclofenac appear to have lower rates
Certain NSAIDs, such as aspirin, have been marketed in enteric-coated formulations that manufacturers claim reduce the incidence of gastrointestinal ADRs. Similarly, some believe that rectal formulations may reduce gastrointestinal ADRs. However, consistent with the systemic mechanism of such ADRs, and in clinical practice, these formulations have not demonstrated a reduced risk of GI ulceration.
Commonly, gastric (but not necessarily intestinal) adverse effects can be reduced through suppressing acid production, by concomitant use of a proton pump inhibitor, e.g., omeprazole, esomeprazole; or the prostaglandin analogue misoprostol. Misoprostol is itself associated with a high incidence of gastrointestinal ADRs (diarrhea). While these techniques may be effective, they are expensive for maintenance therapy.
Inflammatory bowel disease[edit]NSAIDs should be used with caution in individuals with inflammatory bowel disease (e.g., Crohn's disease or ulcerative colitis) due to their tendency to cause gastric bleeding and form ulceration in the gastric lining. Pain relievers such as paracetamol (also known as acetaminophen) or drugs containing codeine (which slows down bowel activity) are safer medications for pain relief in IBD.
Renal
NSAIDs are also associated with a fairly high incidence of renal adverse drug reactions (ADRs). The mechanism of these renal ADRs is due to changes in renal haemodynamics (kidney blood flow), ordinarily mediated by prostaglandins, which are affected by NSAIDs. Prostaglandins normally cause vasodilation of the afferent arterioles of the glomeruli. This helps maintain normal glomerular perfusion andglomerular filtration rate (GFR), an indicator of renal function. This is particularly important in renal failure where the kidney is trying to maintain renal perfusion pressure by elevated angiotensin II levels. At these elevated levels, angiotensin II also constricts the afferent arteriole into the glomerulus in addition to the efferent arteriole it normally constricts. Prostaglandins serve to dilate the afferent arteriole; by blocking this prostaglandin-mediated effect, particularly in renal failure, NSAIDs cause unopposed constriction of the afferent arteriole and decreased RPF (renal perfusion pressure).
Common ADRs associated with altered renal function include:
- Salt (Sodium) and fluid retention
- Hypertension (high blood pressure)
In rarer instances NSAIDs may also cause more severe renal conditions:NSAIDs in combination with excessive use of phenacetin and/or paracetamol (acetaminophen) may lead to analgesic nephropathy.
Photosensitivity
Photosensitivity is a commonly overlooked adverse effect of many of the NSAIDs. The 2-arylpropionic acids are the most likely to produce photosensitivity reactions, but other NSAIDs have also been implicated including piroxicam, diclofenac and benzydamine.
Benoxaprofen, since withdrawn due to its hepatotoxicity, was the most photoactive NSAID observed. The mechanism of photosensitivity, responsible for the high photoactivity of the 2-arylpropionic acids, is the ready decarboxylation of the carboxylic acid moiety. The specific absorbance characteristics of the different chromophoric 2-aryl substituents, affects the decarboxylation mechanism. While ibuprofen has weak absorption, it has been reported as a weak photosensitising agent.
During pregnancy
NSAIDs are not recommended during pregnancy, particularly during the third trimester. While NSAIDs as a class are not directteratogens, they may cause premature closure of the fetal ductus arteriosus and renal ADRs in the fetus. Additionally, they are linked withpremature birth and miscarriage. Aspirin, however, is used together with heparin in pregnant women with antiphospholipid antibodies. Additionally, Indomethacin is used in pregnancy to treat polyhydramnios by reducing fetal urine production via inhibiting fetal renal blood flow.
In contrast, paracetamol (acetaminophen) is regarded as being safe and well-tolerated during pregnancy, but Leffers et al. released a study in 2010 indicating that there may be associated male infertility in the unborn. Doses should be taken as prescribed, due to risk of hepatotoxicity with overdoses.
In France, the country's health agency contraindicates the use of NSAIDs, including aspirin, after the sixth month of pregnancy.
Allergy/allergy-like hypersensitivity reactions. A variety of allergic or allergic-like NSAID hypersensitivity reactions follow the ingestion of NSAIDs. These hypersensitivity reactions differ from the other adverse reactions listed here which are toxicity reactions, i.e. unwanted reactions that result from the pharmacological action of a drug, are dose-related, and can occur in any treated individual; hypersensitivity reactions are idiosyncratic reactions to a drug.
Some NSAID hypersensitivity reactions are truly allergic in origin:
1) repetitive IgE-mediated urticarial skin eruptions,angioedema, and anaphylaxis following immediately to hours after ingesting one structural type of NSAID but not after ingesting structurally unrelated NSAIDs; and
2) Comparatively mild to moderately severe T cell-mediated delayed onset (usually more than 24 hour), skin reactions such as maculopapular rash, fixed drug eruptions, photosensitivity reactions, delayed urticaria, and contact dermatitis; or
3) far more severe and potentially life-threatening t-cell mediated delayed systemic reactions such as the DRESS syndrome,acute generalized exanthematous pustulosis, the Stevens–Johnson syndrome, and toxic epidermal necrolysis. Other NSAID hypersensitivity reactions are allergy-like symptoms but do not involve true allergic mechanisms; rather, they appear due to the ability of NSAIDs to alter the metabolism of arachidonic acid in favor of forming metabolites that promote allergic symptoms.
Afflicted individuals may be abnormally sensitive to these provocative metabolites and/or overproduce them and typically are susceptible to a wide range of structurally dissimilar NSAIDs, particularly those that inhibit COX1. Symptoms, which develop immediately to hours after ingesting any of various NSAIDs that inhibit COX-1, are:
1) exacerbations of asthmatic and rhinitis (see aspirin-induced asthma) symptoms in individuals with a history of asthma or rhinitis and
2) exacerbation or first-time development of wheals and/or angioedema in individuals with or without a history of chronic urticarial lesions or angioedema.
Other
Common adverse drug reactions (ADR), other than listed above, include: raised liver enzymes, headache, dizziness. Uncommon ADRs include: hyperkalaemia, confusion, bronchospasm, rash. Rapid and severe swelling of the face and/or body. Ibuprofen may also rarely cause irritable bowel syndrome symptoms. NSAIDs are also implicated in some cases of Stevens–Johnson syndrome.
Most NSAIDs penetrate poorly into the central nervous system (CNS). However, the COX enzymes are expressed constitutively in some areas of the CNS, meaning that even limited penetration may cause adverse effects such as somnolence and dizziness.
In very rare cases, ibuprofen can cause aseptic meningitis.
As with other drugs, allergies to NSAIDs might exist. While many allergies are specific to one NSAID, up to 1 in 5 people may have unpredictable cross-reactive allergic responses to other NSAIDs as well.
Drug interactions
- NSAIDs reduce renal blood flow and thereby decrease the efficacy of diuretics, and inhibit the elimination of lithium and methotrexate.
- NSAIDs cause hypocoagulability, which may be serious when combined with other drugs that also decrease blood clotting, such aswarfarin.[51]
- NSAIDs may aggravate hypertension (high blood pressure) and thereby antagonize the effect of antihypertensives, such as ACE Inhibitors.
- NSAIDs may interfere and reduce efficiency of SSRI antidepressants.
Mechanism of action
Most NSAIDs act as nonselective inhibitors of the enzyme cyclooxygenase (COX), inhibiting both the cyclooxygenase-1 (COX-1) and cyclooxygenase-2 (COX-2) isoenzymes. This inhibition is competitively reversible (albeit at varying degrees of reversibility), as opposed to the mechanism of aspirin, which is irreversible inhibition. COX catalyzes the formation of prostaglandins and thromboxane fromarachidonic acid (itself derived from the cellular phospholipid bilayer by phospholipase A2). Prostaglandins act (among other things) as messenger molecules in the process of inflammation. This mechanism of action was elucidated by John Vane (1927–2004), who received a Nobel Prize for his work (see Mechanism of action of aspirin).
COX-1 is a constitutively expressed enzyme with a "house-keeping" role in regulating many normal physiological processes. One of these is in the stomach lining, where prostaglandins serve a protective role, preventing the stomach mucosa from being eroded by its own acid. COX-2 is an enzyme facultatively expressed in inflammation, and it is inhibition of COX-2 that produces the desirable effects of NSAIDs.
When nonselective COX-1/COX-2 inhibitors (such as aspirin, ibuprofen, and naproxen) lower stomach prostaglandin levels, ulcers of thestomach or duodenum internal bleeding can result.
NSAIDs have been studied in various assays to understand how they affect each of these enzymes. While the assays reveal differences, unfortunately different assays provide differing ratios.
The discovery of COX-2 led to research to development of selective COX-2 inhibiting drugs that do not cause gastric problems characteristic of older NSAIDs.
Paracetamol (acetaminophen) is not considered an NSAID because it has little anti-inflammatory activity. It treats pain mainly by blocking COX-2 mostly in the central nervous system, but not much in the rest of the body.
However, many aspects of the mechanism of action of NSAIDs remain unexplained, and for this reason further COX pathways are hypothesized. The COX-3 pathway was believed to fill some of this gap but recent findings make it appear unlikely that it plays any significant role in humans and alternative explanation models are proposed.
NSAIDs are also used in the acute pain caused by gout because they inhibit urate crystal phagocytosis besides inhibition of prostaglandin synthase.
Antipyretic activity
- NSAIDS have antipyretic activity and can be used to treat fever. Fever is caused by elevated levels of prostaglandin E2, which alters the firing rate of neurons within the hypothalamus that control thermoregulation.
- Antipyretics work by inhibiting the enzyme COX, which causes the general inhibition of prostanoid biosynthesis (PGE2) within the hypothalamus.
- PGE2 signals to the hypothalamus to increase the body's thermal set point. Ibuprofen has been shown more effective as an antipyretic thanparacetamol (acetaminophen).
- Arachidonic acid is the precursor substrate for cyclooxygenase leading to the production of prostaglandins F, D & E.
NSAIDs can be classified based on their chemical structure or mechanism of action. Older NSAIDs were known long before their mechanism of action was elucidated and were for this reason classified by chemical structure or origin. Newer substances are more often classified by mechanism of action.
Salicylates
- Aspirin (acetylsalicylic acid)
- Diflunisal (Dolobid)
- Salicylic acid and other salicylates
- Salsalate (Disalcid)
- indomethacin
- Tolmetin
- Sulindac
- Etodolac
- Ketorolac
- Diclofenac
- Aceclofenac
- Nabumetone (drug itself is non-acidic but the active, principal metabolite has a carboxylic acid group)
- Piroxicam
- Meloxicam
- Tenoxicam
- Droxicam
- Lornoxicam
- Isoxicam (withdrawn from market 1985)
- Phenylbutazone (Bute)
- Celecoxib (FDA alert)
- Rofecoxib (withdrawn from market)
- Valdecoxib (withdrawn from market)
- Parecoxib FDA withdrawn, licensed in the EU
- Lumiracoxib TGA cancelled registration
- Etoricoxib not FDA approved, licensed in the EU
- Firocoxib used in dogs and horses
- Nimesulide (systemic preparations are banned by several countries for the potential risk of hepatotoxicity)
- Clonixin
- Licofelone acts by inhibiting LOX (lipooxygenase) & COX and hence known as 5-LOX/COX inhibitor
- H-harpagide in Figwort or Devil's Claw
Most NSAIDs are chiral molecules (diclofenac is a notable exception). However, the majority are prepared in a racemic mixture.
Typically, only a single enantiomer is pharmacologically active.
For some drugs (typically profens), an isomerase enzyme in vivo converts the inactive enantiomer into the active form, although its activity varies widely in individuals.
This phenomenon is likely responsible for the poor correlation between NSAID efficacy and plasma concentration observed in older studies, when specific analysis of the active enantiomer was not performed.
Ibuprofen and ketoprofen are now available in single, active enantiomer preparations (dexibuprofen and dexketoprofen), which purport to offer quicker onset and an improved side-effect profile. Naproxen has always been marketed as the single active enantiomer.
Main practical differences.
NSAIDs within a group tend to have similar characteristics and tolerability. There is little difference in clinical efficacy among the NSAIDs when used at equivalent doses. Rather, differences among compounds usually relate to dosing regimens (related to the compound'selimination half-life), route of administration, and tolerability profile.
Regarding adverse effects, selective COX-2 inhibitors have lower risk of gastrointestinal bleeding, but a substantially more increased risk of myocardial infarction than the increased risk from nonselective inhibitors. Some data also supports that the partially selectivenabumetone is less likely to cause gastrointestinal events. The nonselective naproxen appears risk-neutral with regard to cardiovascular events.
A consumer report noted that ibuprofen, naproxen, and salsalate are less expensive than other NSAIDs, and essentially as effective and safe when used appropriately to treat osteoarthritis and pain.
Pharmacokinetics[edit]Most nonsteroidal anti-inflammatory drugs are weak acids, with a pKa of 3–5. They are absorbed well from the stomach and intestinal mucosa. They are highly protein-bound in plasma (typically >95%), usually to albumin, so that their volume of distribution typically approximates to plasma volume. Most NSAIDs are metabolised in the liver by oxidation and conjugation to inactive metabolites that typically are excreted in the urine, though some drugs are partially excreted in bile. Metabolism may be abnormal in certain disease states, and accumulation may occur even with normal dosage.
Ibuprofen and diclofenac have short half-lives (2–3 hours). Some NSAIDs (typically oxicams) have very long half-lives (e.g. 20–60 hours).
History
One of the first advertisements for Bayer Aspirin, published in The New York Times in 1917From the era of Greek medicine to the mid-19th century, the discovery of medicinal agents was classed as an empirical art; folklore and mythological guidance were combined in deploying the vegetable and mineral products that made up the expansive pharmacopoeia of the time. Myrtleleaves were in use by 1500 BCE. Hippocrates (460–377 BCE) first reported using willow bark[76] and in 30 BCE Celsus described the signs of inflammation and also used willow bark to mitigate them. On 25 April 1763, Edward Stone wrote to the Royal Society describing his observations on the use of willow bark-based medicines in febrile patients.[77] The active ingredient of willow bark, a glycosidecalled salicin, was first isolated by Johann Andreas Buchner in 1827. By 1829, French chemist Henri Leroux had improved the extraction process to obtain about 30g of purified salicin from 1.5 kg of bark.[77]
By hydrolysis, salicin releases glucose and salicylic alcohol which can be converted into salicylic acid, both in vivo and through chemical methods.[76] The acid is more effective than salicin and, in addition to its fever-reducing properties, is anti-inflammatory and analgesic. In 1869, Hermann Kolbesynthesised salicylate, although it was too acidic for the gastric mucosa.[76] The reaction used to synthesise aromatic acid from a phenol in the presence of CO2 is known as the Kolbe-Schmitt reaction.[78][79][80]
By 1897 the German chemist Felix Hoffmann and the Bayer company prompted a new age of pharmacology by converting salicylic acid into acetylsalicylic acid—named aspirin by Heinrich Dreser. Other NSAIDs were developed from the 1950s forward.[77] In 2001, NSAIDs accounted for 70,000,000 prescriptions and 30 billion over-the-counter doses sold annually in the United States.[16]
Veterinary use[edit]Research supports the use of NSAIDs for the control of pain associated with veterinary procedures such as dehorning and castration of calves. The best effect is obtained by combining a short-term local anesthetic such as lidocaine with an NSAID acting as a longer term analgesic. However, as different species have varying reactions to different medications in the NSAID family, little of the existing research data can be extrapolated to animal species other than those specifically studied, and the relevant government agency in one area sometimes prohibits uses approved in other jurisdictions.
For example, ketoprofen's effects have been studied in horses more than in ruminants but, due to controversy over its use in racehorses, veterinarians who treat livestock in the United States more commonly prescribe flunixin meglumine, which, while labeled for use in such animals, is not indicated for post-operative pain.